Interfaces play important role in condensate behavior
Research from labs of Rohit Pappu, collaborators sheds light on condensate characteristics
Before mixing an oil-and-vinegar-based salad dressing, the individual drops of vinegar are easily seen suspended in the oil, each with a perfectly circular boundary that delineates the two liquids. In the same way, our cells contain condensed bundles of proteins and nucleic acids called condensates delineated by clear boundaries. The boundaries are known as interfaces and given that condensates talk to one another through their interfaces, the structural features of the interface are of significant interest. New research has uncovered unique features of interfaces of model condensates. The findings are relevant because the features interfaces are relevant to the seeding of fibrillar conformations that are associated with neurodegenerative diseases such as amyotrophic lateral sclerosis (ALS).
A team of researchers led by Rohit Pappu, the Gene K. Beare Distinguished Professor and director of the Center for Biomolecular Condensates in the McKelvey School of Engineering at Washington University in St. Louis, recently focused on defining the molecular scale features of interfaces of condensates. The research, driven by Mina Farag, an MD/PhD student in the Pappu lab and first author of the paper, were generated in collaboration with Tanja Mittag and her lab at St. Jude Children’s Research Hospital. The results were published in Nature Communications Dec. 13, 2022.
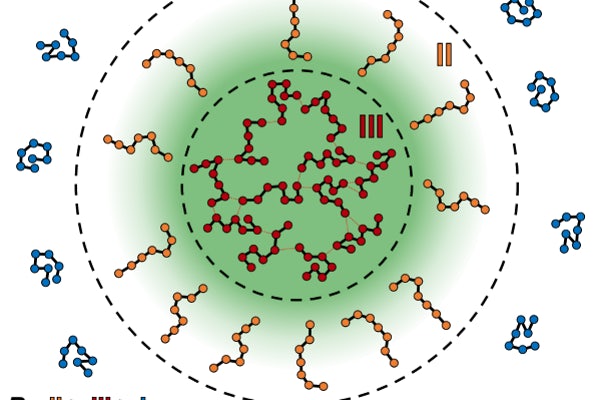
A surprising finding was that interfaces, which might seem to be uniform and infinitesimally thin from the vinegar in oil picture, are thick and defined by specific shape and size preferences at the molecular level, Pappu said. Within condensates, the molecules are organized into a small-world structure that we readily recognize from the hub-and-spoke network structure of airports for commercial airlines.
“There is a particular type of connectivity that defines the way these molecules are organized, and that is because they have viscoelastic properties that makes them either elastic on short timescales and viscous on long timescales, much like putty,” Pappu said.
As the protein molecules go through the interface, the shapes and sizes of the molecules change in a way that these are unique to the interface, the team’s research found.
“We made a very striking observation that the conformations, or the shapes, of these molecules were very distinctive as they go through the interface, and those types of conformations poise them to be reactive,” Pappu said. “That can be good for facilitating biochemical reactions inside a cell, or be deleterious in the context of ALS, whereby the interface catalyzes the fibrillar growth in motor neurons.”
In Pappu’s lab, Farag used data from Mittag’s lab to train a machine learning model that describes the interactions among the molecules. This allowed them to simulate condensate formation in a computer. The simulations reproduce condensation for 30 different variants of a specific protein domain that is associated with ALS. Importantly, the simulation paradigm affords a way to design proteins with bespoke condensate structures and interfacial properties.
“We think that the distinct conformational preferences in interfaces contribute to low interfacial tensions of condensates. However, the specific chemistries of different interfaces are likely to enable functional selectivity. This seems like a very happy medium between physics and chemistry, as gleaned using engineering-based methods.”
Farag M, Cohen SR, Borcherds WM, Bremer A, Mittag T, Pappu RV. Condensates formed by prion-like low-complexity domains have small-world network structures and interfaces defined by expanded conformations. Nature Communications. Published online Dec. 13, 2022. https://doi.org/10.1038/s41467-022-35370-7
This research was supported by the National Institutes of Health (R01NS121114); the St. Jude Children’s research collaborative on the biophysics and biology of RNP granules; and the Air Force Office of Scientific Research (FA9550-20-1-0241).





