Timing matters: Condensates’ composition determined by when RNA is added
Rohit Pappu, collaborators from Duke University make discovery with model fungus
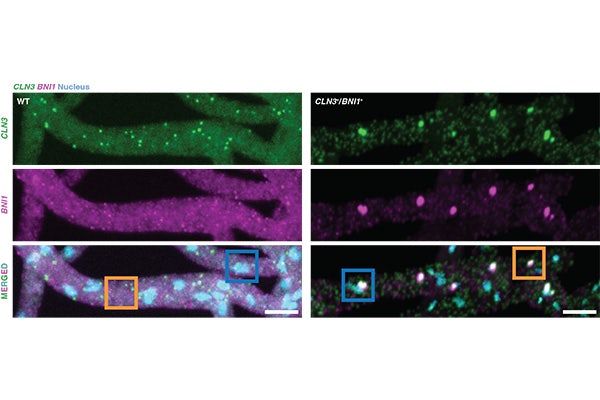
Biomolecular condensates transport RNA molecules inside of cells for functions such as cell signaling and regulating cell processes, but little is known about how they form distinct compositional identities, similar to how oil and water stay separated.
Rohit V. Pappu, the Gene K. Beare Distinguished Professor of biomedical engineering in the McKelvey School of Engineering at Washington University in St. Louis, in collaboration with Amy S. Gladfelter, professor of cell biology and of biomedical engineering at Duke University, and their labs have found that the order that different RNA molecules are added to the condensates determines how they will be composed, providing new information on how these important cellular compartments are formed. Pappu also is director of the Center for Biomolecular Condensates in the McKelvey School of Engineering, and Gladfelter is a member of the center’s Scientific Advisory Committee.
The team made the discovery using a protein from Ashbya gossypii, a fungus with long filaments that is closely related to yeast. Using live cells, they found that a lack of dynamical control, achieved by synchronizing the production of RNA molecules, resulted in a loss of compositional identity of condensates, which impacted the ability of the fungi to form long, healthy cells with well-defined branches. These branches allow cells to optimize growth in specific areas, so cells can explore and extract resources from the environment, and they are made possible by the ability to package RNA molecules and localize to sites where they are needed, Pappu said.
“Condensates are the vehicles for regulation and transport of RNA materials, and ensuring that compositional identity is maintained is key,” Pappu said. “That the interactions involving RNA molecules are strong enough to ensure that compositional identity can be achieved by asynchronous production of RNA is quite remarkable, because it implies that time becomes a whole new dimension for us to explore when thinking about condensate formation and the generation and maintenance of compositional identity.”
“Time offers such a powerful but often overlooked mechanism of control over molecular behavior, Gladfelter said. “We often focus on molecular details as drivers of specificity in how complexes in cells assemble. But what this work revealed is how timing of appearance of molecules can be just as potent in informing what molecules come together when in a cell.”
Previously, researchers thought that sequence-specific interactions between the Whi3 protein from Ashbya gossypii and different RNA molecules provided spontaneous control over separating the components in the condensates and compositional identity. In the new research, the Pappu and Gladfelter teams used theory and computations to create rules to analyze the data from their experiments.
“It turned out that the rules that govern spontaneous control over compositional identity are not sufficient to explain the behavior of these systems,” Pappu said. “Instead, timing matters: The order in which different RNA molecules are added to the mixture determines whether a ternary system will be well-mixed or compositionally distinct. Our studies identified the features of RNA molecules that contribute to dynamical control over compositional identity of condensates.”
Going forward, the team will look at whether compositional identity of condensates, realized by dynamical control, can be regulated by active processes and if the crosstalk between condensates is also under dynamical control. Answers to these questions can help researchers understand how condensates contribute to the array of functions that are influenced by fungi as well as understand condensate biology in axons, cardiac myocytes and other cell types where spatial and temporal control are required.
“If we understand how dynamical control over compositional identity is achieved, then we can find ways to engineer cells, specifically fungi, to achieve functionally relevant, and compositionally distinct condensates as novel materials that perform bespoke functions in desired spaces at desired times — the dream of synthetic biology,” Pappu said.
Lin AZ, Ruff KM, Dar F, Jalihal A, King MR, Lalmansingh JM, Posey AE, Erkamp NA, Seim I, Gladfelter AS, Pappu RV. Dynamical control enables the formation of demixed biomolecular condensates. Nature Communications, Nov. 24, 2023, DOI: 10.1038/s41467-023-43489-4.
The latest, public release of LaSSI, developed and maintained by the Pappu lab, is available on the Pappu lab Github repository at https://github.com/Pappulab/LASSI.
This work was supported by grants from the Air Force Office of Scientific Research (FA9550-20-1-0241), the St. Jude Research Collaborative on the Biology and Biophysics of RNP granules, and the National Institutes of Health (F32GM146418-01A1, R01NS121114).





