New imaging technology may reduce surgeries for rectal cancer patients
Quing Zhu, Matthew Mutch pursue dual modality photoacoustic and ultrasound imaging, deep learning method as ‘a major step forward’
Colorectal cancer is the third most common cause of cancer death in the United States among men and women, and the incidence among people under age 50 has risen to one in five new diagnoses, according to the American Cancer Society. While treatment allows some patients to avoid surgery, existing technology makes it difficult to determine whether the cancer has been successfully treated with no residual cancers.
Quing Zhu, a biomedical engineer in the McKelvey School of Engineering, and Matthew Mutch, MD, a colorectal surgeon at the School of Medicine, both at Washington University in St. Louis, and their collaborators have been working together to address this problem by developing a new imaging technology combining photoacoustic microscopy, ultrasound and deep learning to better determine whether a rectal cancer patient is successfully treated with radiation and chemotherapy and can be safely followed-up with nonsurgical imaging monitoring. With a four-year, $1.75 million grant from the National Institutes of Health, the team led by Zhu and Mutch will pursue development of this new technology that would help physicians to accurately identify a treated rectal tumor bed with residual cancers that need surgery or normalized rectal tissue without need for surgery.
In the past, Stage 2 and 3 rectal cancers have been treated with radiation and chemotherapy followed by surgical removal of the cancerous tissue. However, advances in preoperative treatment enable up to 35% of these patients to achieve complete tumor death with radiation and chemotherapy alone. In these individuals, surgical resection has shown no benefit and carries the significant risks of major complications, prolonged recovery and reduced quality of life. Existing imaging modalities to determine whether the tumor has been eliminated make it difficult for surgeons to distinguish between a residual cancer and scar tissue, resulting in surgery remaining the standard of care.
“Improved imaging methods could resolve this problem and allow widespread adoption of nonoperative management called ‘watch and wait’ for hundreds of thousands of rectal cancer patients,” said Zhu, a pioneer in combining ultrasound and other optical imaging modalities to better detect cancers. “If successful, this technology will directly reduce the number of unnecessary surgeries for rectal cancer and improve the patients’ quality of life.”
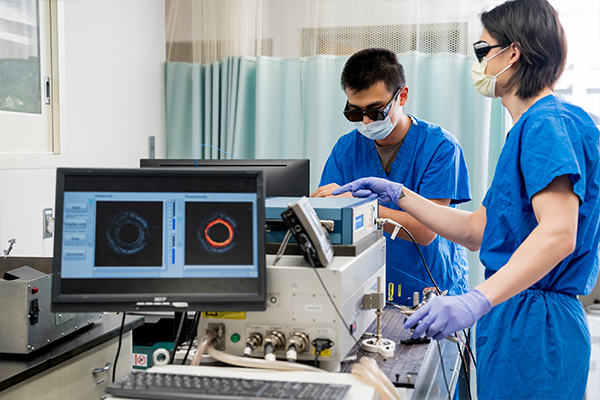
Zhu, the Edwin H. Murty Professor of Engineering; former doctoral associate Xiandong Leng; and current doctoral students in her lab Sitai Kou, Yixiao Lin, Taylor Nie, Sanskar Thakur and Yitian Zhang have developed a co-registered acoustic resolution photoacoustic microscopy and ultrasound (AR-PAM/US) endoscopy prototype system and neural network classifiers that they expect to perform better than standard-of-care practices at determining whether the cancer has been eliminated from rectal tissue. Her team first tested the system in a feasibility study in 2019-2021 and found that their method was better able to determine the presence of residual tumors in treated rectal tissues than other types of imaging, such as MRI.
With the new funding, the team will prospectively assess the ability of the technology to improve standard-of-care imaging in a group of rectal cancer patients who don’t know whether they have a residual tumor or scar tissue. In addition, the team will monitor a group of patients to determine if the AR-PAM/US technology can assess changes in tumor vascular and blood oxygen saturation and diagnose the response of the tumor, both during and after treatment.
Photoacoustic imaging uses a laser to excite tissue. The laser light absorption causes emission of sound waves that reveal properties of the tissue vasculature, a biomarker for cancerous tissue due to tumor angiogenesis. Zhu’s team is constructing and optimizing new probes that use AR-PAM/US to efficiently assess the rectum after treatment.
Zhu and her team have built a custom pattern-recognition neural network that provides a quantitative interpretation of the AR-PAM and US images and is expected to help surgeons make real-time diagnoses. In their previous study, the imaging and neural network showed that tumor beds in rectal tissue revert from patterns that represent cancer to those that are typical of a normal rectum, showing a complete response to treatment. They expect that their AR-PAM/US system and the neural net classifiers will allow surgeons to example the tumor’s microvessel network and assess treatment response after the radiation and chemotherapy alone and that their studies will perform better than standard-of-care methods to predict a patient’s response to treatment both at the end of treatment and during the “watch-and-wait” period for up to two years after treatment.
Zhu is collaborating with School of Medicine clinicians Mutch, the Solon and Bettie Gershman Chair in Colon and Rectal Surgery, chief of the Section of Colon and Rectal Surgery and professor of surgery; Steven Hunt, MD, associate professor of surgery; William Chapman Jr., MD, who will join the School of Medicine in August as an assistant professor; Anup Shetty, MD, assistant professor of radiology; Pooja Navale, MD, assistant professor of pathology and immunology; Hyun Kim, MD, assistant professor of radiation oncology; and Fei Wan, assistant professor of public health. The pilot study was funded by the Siteman Cancer Center Pre-R01 from 2020-2022.
This technology and study are integral to improving the care of patients with rectal cancer, Mutch said.
“There has been a significant shift in the treatment paradigm for rectal cancer, he said. “Historically all patients got chemoradiation therapy followed by surgery. However, now we have improved the results where up to one-third of patients ultimately can avoid surgery. The intent of photoacoustic imaging is to help us better differentiate those that have had a complete response to their neoadjuvant treatment versus those that have residual tumor. Being able to identify this as early as possible greatly impacts the overall outcome, and photoacoustic imaging will provide a major step forward in this process.”





